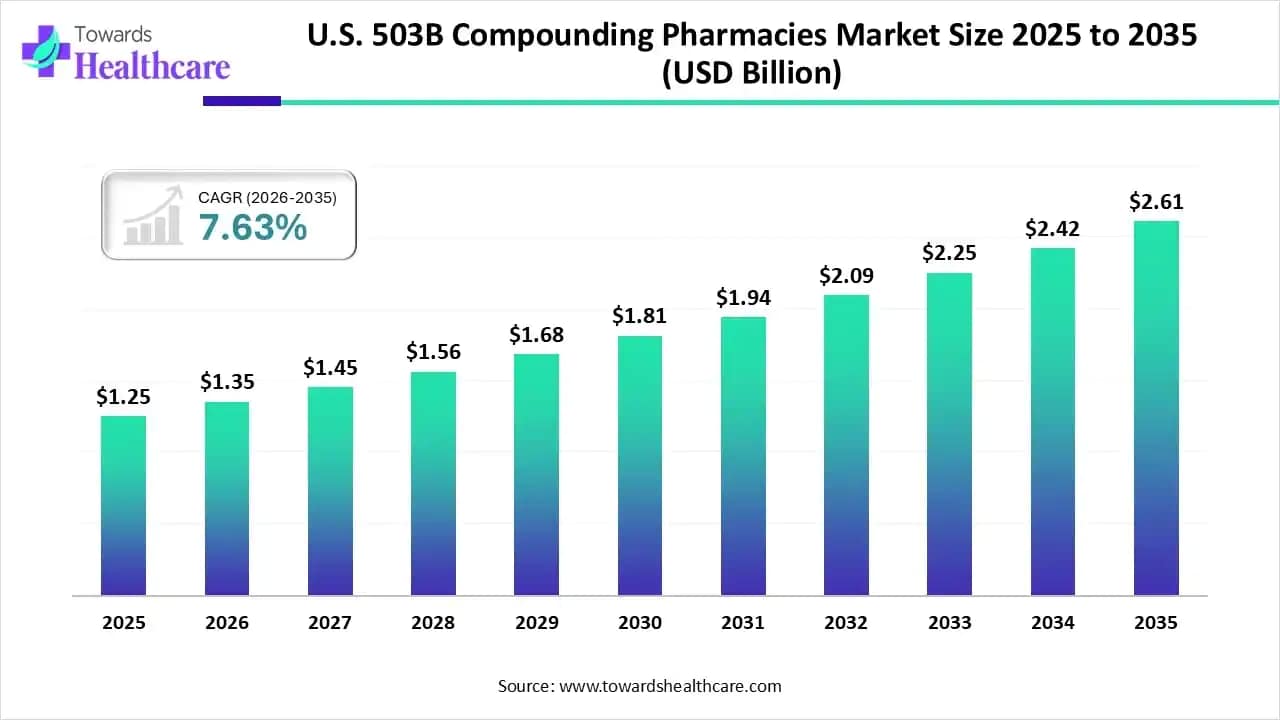
Ottawa, April 17, 2026 (GLOBE NEWSWIRE) -- The U.S. 503B compounding pharmacies market size is calculated at USD 1.35 billion in 2026 and is expected to reach around USD 2.61 billion by 2035, growing at a CAGR of 7.63% for the forecasted period.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/5386
The 503B compounding pharmacies are also referred to as compounding outsourcing facilities, which are specialized types of drug manufacturing organizations that are critical to patient care. These are 503B outsource compounding centers that maintain a balance between traditional compounding pharmacies and large pharmaceutical manufacturers. Patients who may not be able to use commercially available formulations due to allergies, dosing requirements, or rare diseases generally prefer compounding that provides them access to medications.
According to the current good manufacturing practices (cGMP), compounded drugs are manufactured at registered and suitable facilities by using properly calibrated equipment. The manufacturers prefer properly validated production systems and use only authorized materials. These materials are processed by trained personnel, and all products are packaged and labelled based on a quality management system. On the other hand, the specialized services and solutions include API-to-sterile, sterile-to-sterile, biologics, controlled substances, OR anesthesia, ophthalmics, pain management, etc.
The U.S. 503B Compounding Pharmacies Market: Highlights
- U.S. 503B compounding pharmacies industry poised to reach USD 1.35 billion by 2026.
- Forecasted to grow to USD 2.61 billion by 2035.
- Expected to maintain a CAGR of 7.63% from 2026 to 2035.
- The Northeast region held a major revenue share of the market in 2025.
- The South region is expected to witness the fastest growth during the predicted timeframe.
- By product type, the sterile compounded drugs segment dominated the market share by 68% in 2025.
- By product type, the non-sterile compounded drugs segment is expected to show the fastest growth in the forecast period.
- By formulation type, the injections & infusions segment accounted for the highest revenue share of the U.S. 503B compounding pharmacies market by 38% in 2025.
- By formulation type, the capsules & tablets segment is expected to grow at the fastest CAGR in the market during the forecast period.
- By therapeutic application, the pain management segment led the market share by 24% in 2025.
- By therapeutic application, the oncology segment is expected to grow with the highest CAGR in the market during the studied years.
- By compounding type, the batch compounding segment registered its dominance over the global market share by 64% in 2025.
- By compounding type, the patient-specific compounding segment is projected to expand significantly in the market in the coming years.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Market Overview
Broad Reach of 503B Outsourcing Facilities
The U.S. 503B compounding pharmacies market is driven by several state and local regulatory bodies to harmonize safe and effective drug manufacturing. The products provided by these outsourcing facilities pass rigorous testing of potency, sterility, and quality. The 503B compounding pharmacies support the regulatory requirements of ambulatory surgical centers, hospitals, and other health systems. These facilities need to comply with numerous requirements of traditional pharmaceutical manufacturers such as ISO, FDA, cGMP, DQSA, and USP regulations.
Top 10 Most Commercialized APIs by 503B Outsourcing Facilities
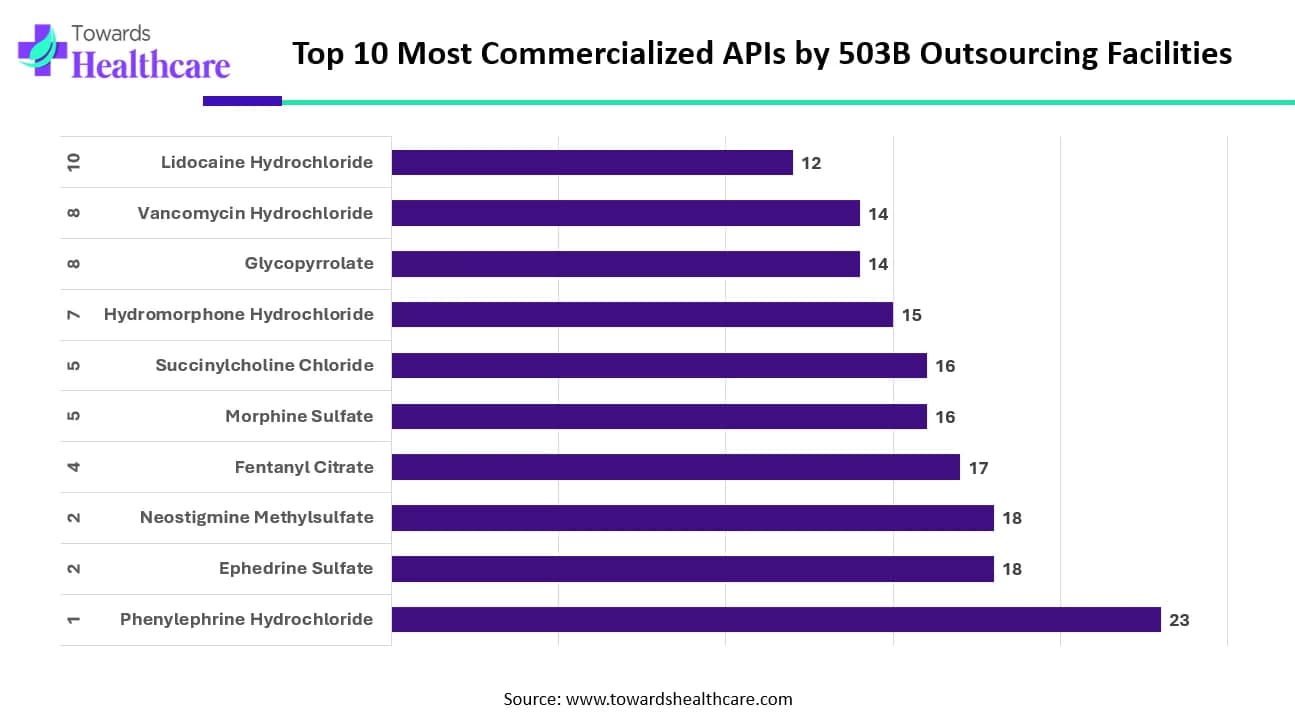
The data highlights which active pharmaceutical ingredients (APIs) are most commonly commercialized by 503B outsourcing facilities. Phenylephrine Hydrochloride leads the list, with the highest number of facilities producing products that contain it. This strong demand reflects its widespread use in emergency and critical care settings.
Ephedrine Sulfate and Neostigmine Methylsulfate follow closely, showing equal levels of commercial interest. Their high usage indicates their importance in anesthesia and perioperative care. Fentanyl Citrate and Morphine Sulfate also appear prominently because healthcare providers rely heavily on these analgesics to manage severe pain.
Succinylcholine Chloride and Hydromorphone Hydrochloride remain key products as well, demonstrating the consistent need for neuromuscular blockers and strong opioids in hospital environments. Meanwhile, Glycopyrrolate and Vancomycin Hydrochloride rank slightly lower but still show strong commercialization due to their essential roles in infection control and surgical preparation.
Lidocaine Hydrochloride closes the list but remains a widely used local anesthetic across multiple clinical procedures. Overall, the ranking reflects real-world clinical demand and the continuing reliance of healthcare systems on these critical medications.
The U.S. 503B Compounding Pharmacies Market: Regional Analysis
The Northeast region dominated the market in 2025, owing to persistent drug shortages, growing demand for personalized medicine, technological innovations, hospital outsourcing, and regulatory support. There is an increased trend of hospital outsourcing, expansion of infrastructure and facilities, and shortages of commercially manufactured drugs such as emergency room medications and oncology. Researchers are focusing on specialized therapeutic areas such as oncology, pain management, and the demand for GLP-1.
The South region is expected to witness the fastest growth during the predicted timeframe in the U.S. 503B compounding pharmacies market due to demand for sterile compounding, growing trends of hospital outsourcing, and the integration of AI-powered software and automated compounding systems. There is a growing demand for hormone replacement therapy and personalized medications, and innovations in specialized medicine. Southern facilities are increasingly adopting robotics, automation, and AI-powered workflows to improve regulatory compliance and safety with current good manufacturing practices.
Become a valued research partner with us - https://www.towardshealthcare.com/schedule-meeting
The U.S. 503B Compounding Pharmacies Market: Segmentation Analysis
Product Type Insights
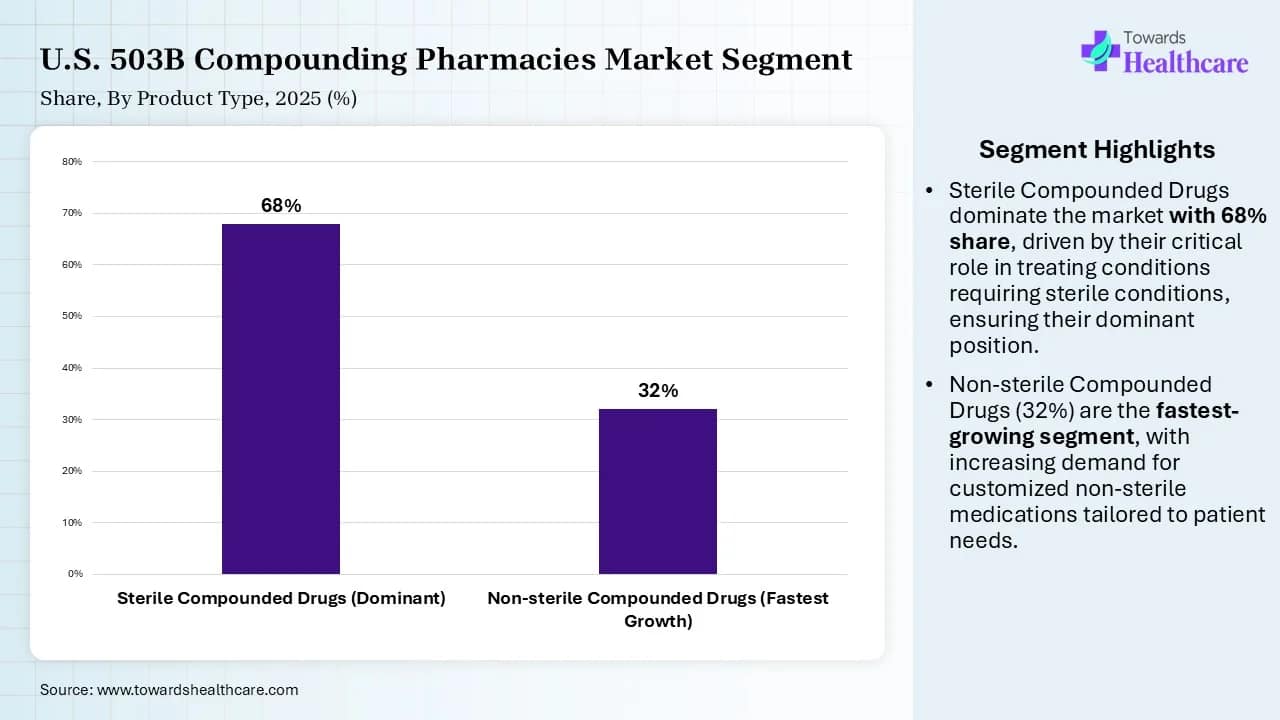
| Product Type | Market Share (%) | |
| Sterile Compounded Drugs (Dominant) | 68 | % |
| Non-sterile Compounded Drugs (Fastest Growth) | 32 | % |
The sterile compounded drugs segment dominated the market share by 68% in 2025, owing to ready-to-use formulations, increased focus on mitigating drug shortages, and compliance with current good manufacturing practices. These drugs have a strategic impact on healthcare providers, which improves supply chain resilience, efficiency, cost, and risk management. These drug manufacturing facilities comply with current good manufacturing practices and need to register with the U.S. Food and Drug Administration (FDA).
The non-sterile compounded drugs segment is expected to show the fastest growth in the U.S. 503B compounding pharmacies market forecast period due to their importance as scalable patient solutions and their impact on quality assurance standards. They are expanding in the form of oral and nasal formulations in the market due to their non-invasive nature and convenience. The 503B facilities provide homogenous and large batches of non-sterile medications such as topical creams, oral liquids, and capsules.
Formulation Type Insights
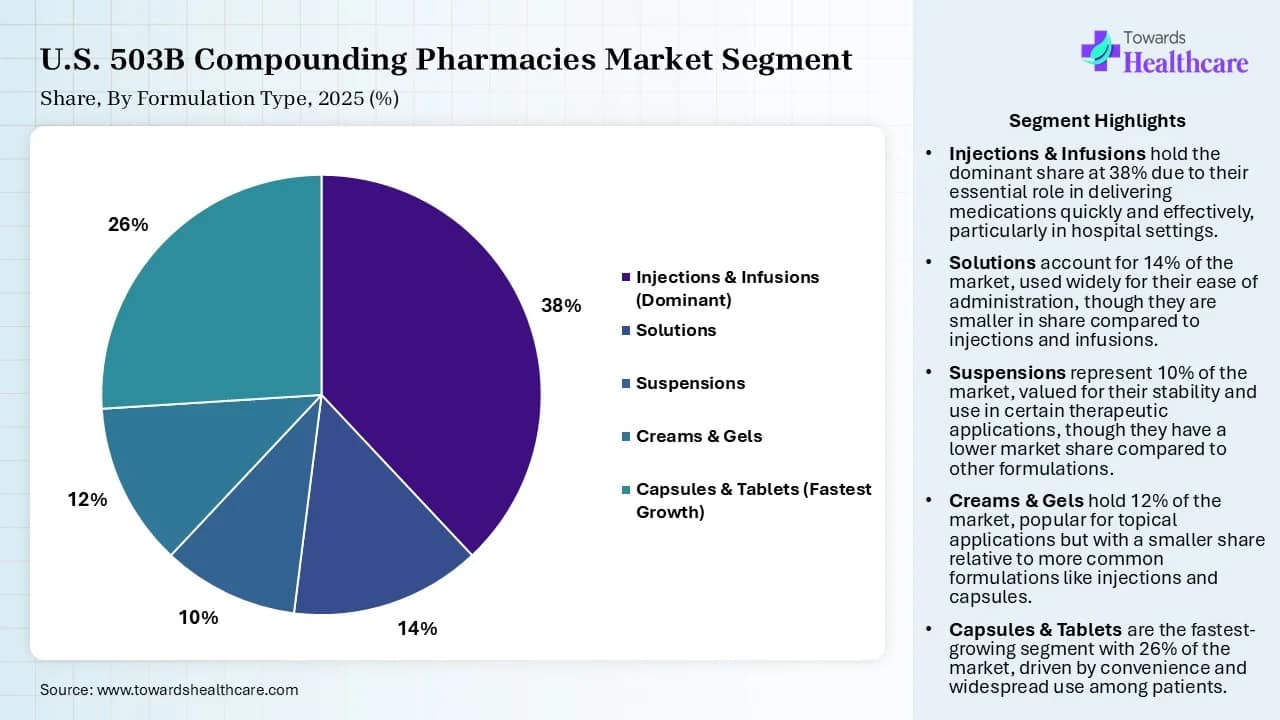
| Formulation Type | Market Share (%) | |
| Injections & Infusions (Dominant) | 38 | % |
| Solutions | 14 | % |
| Suspensions | 10 | % |
| Creams & Gels | 12 | % |
| Capsules & Tablets (Fastest Growth) | 26 | % |
The injections & infusions segment accounted for a dominating position by 38% share in the market in 2025, owing to their importance as ready-to-use injectable medications for clinics and hospitals, specifically during drug shortages. The 503B outsourcing facilities offer injectables as the largest provided services. These services include pre-filled syringes, ready-to-administer IV bags, and other sterile preparations.
The capsules & tablets segment is expected to grow at the fastest CAGR in the U.S. 503B compounding pharmacies market during the forecast period due to the key role of 503B facilities in formulating capsules and tablets without requiring dyes, fillers, or binding agents. These facilities also convert tablets into capsules to improve palatability. The tablets and capsules are playing a more vital role than sterile injectables.
Get the latest insights on life science industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership
Therapeutic Application Insights
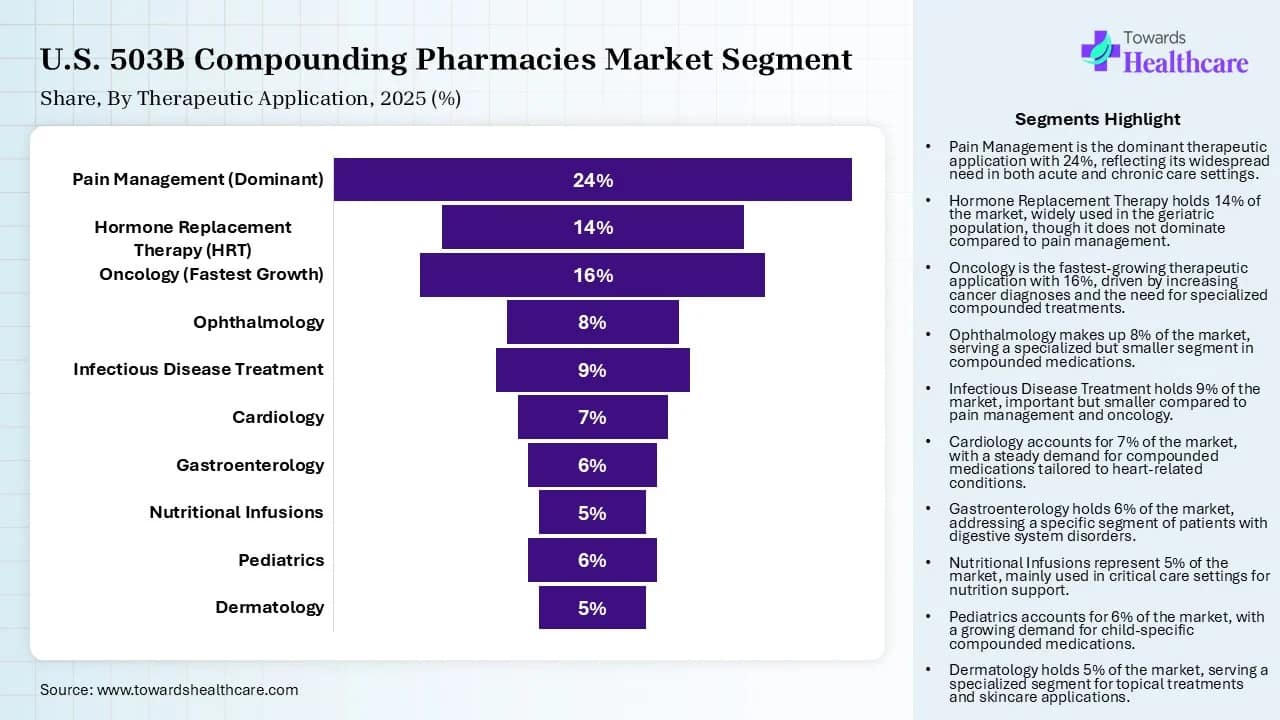
| Therapeutic Application | Market Share (%) | |
| Pain Management (Dominant) | 24 | % |
| Hormone Replacement Therapy (HRT) | 14 | % |
| Oncology (Fastest Growth) | 16 | % |
| Ophthalmology | 8 | % |
| Infectious Disease Treatment | 9 | % |
| Cardiology | 7 | % |
| Gastroenterology | 6 | % |
| Nutritional Infusions | 5 | % |
| Pediatrics | 6 | % |
| Dermatology | 5 | % |
The pain management segment led the market by 24% share in 2025, owing to the large-scale supply of critical analgesics, increased focus on addressing drug shortages, and a shift towards ready-to-use formulations. The 503B facilities offer alternative dosage forms for patients who cannot swallow pills. They also create alternate delivery methods like liquid suspensions and transdermal gels.
The oncology segment is expected to grow with the highest CAGR in the U.S. 503B compounding pharmacies market during the studied years due to the immense role of 503B outsourcing facilities in providing ready-to-administer syringes and enhancing precision dosing. These products, such as infusion bags and pre-filled syringes, improve clinical workflow and reduce medication errors. These facilities also produce tailored dosage forms such as allergen-free versions or specific concentrations that are safe for pediatric patients.
Compounding Type Insights
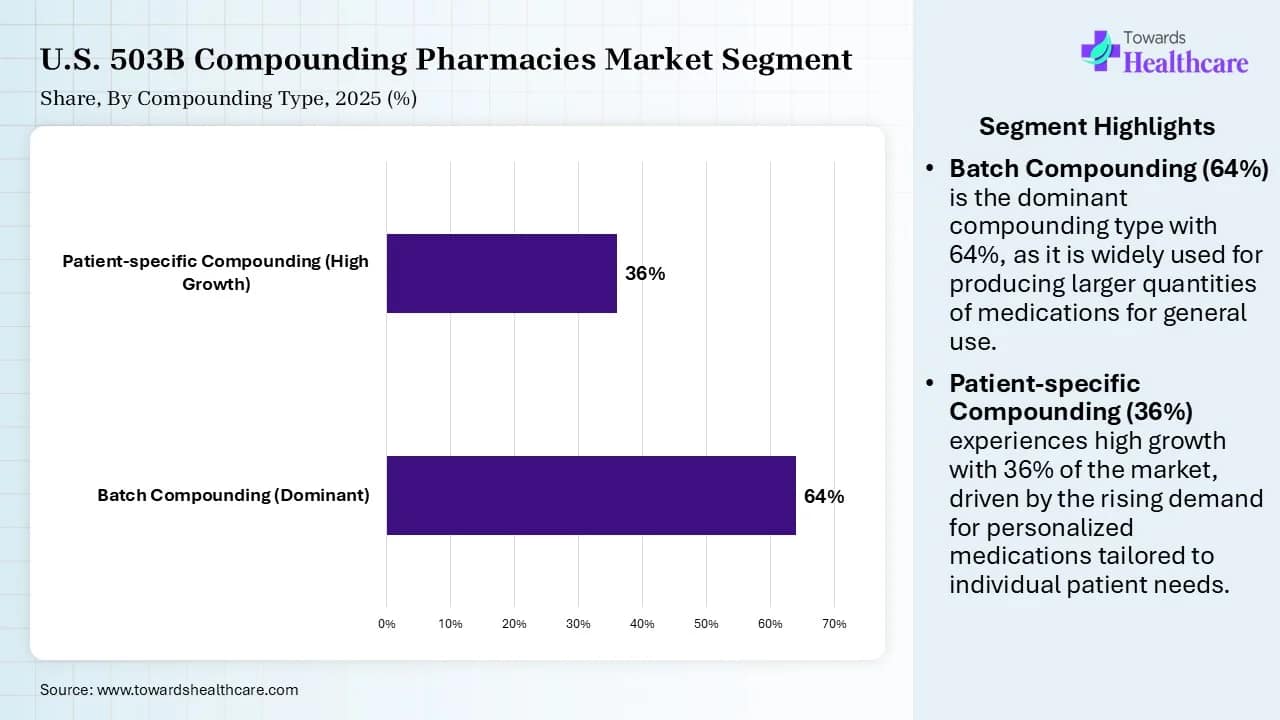
| Compounding Type | Market Share (%) | |
| Batch Compounding (Dominant) | 64 | % |
| Patient-specific Compounding (High Growth) | 36 | % |
The batch compounding segment registered its dominance in the market by 64% share in 2025, owing to the permitted 503B outsourcing facilities to compound copies of FDA-approved drugs by addressing drug shortages. These facilities focus on utilizing automation while producing large batches to reduce per-unit costs for clinics and hospitals. They produce custom formulations with different dosage forms, specific strengths, or those free from preservatives or certain allergens.
The patient-specific compounding segment is projected to expand significantly in the U.S. 503B compounding pharmacies market in the coming years due to the increased focus of businesses on patient-centered dispensing of bulk orders of drugs. The 503B facilities are important in meeting demands for compounding medications that are listed on the drug shortage list of the U.S. FDA. The 503B facilities supply ready-to-use sterile and non-sterile preparations to clinics, hospitals, and ambulatory surgery centers.
Get the latest insights on life science industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership
The U.S. 503B Compounding Pharmacies Market Companies
- Athenex, Inc.
- Wells Pharma of Houston LLC
- SCA Pharma
- Nephron Pharmaceuticals Corporation
- Olympia Pharmacy
- Fagron Compounding Pharmacies
- QuVa Pharma
- Empower Pharmacy
- Wells Pharma of Houston, LLC
- Central Admixture Pharmacy Services, Inc.
- US Compounding Inc.
- ImprimisRx
- Revelation Pharma
- Central Admixture Pharmacy Services
Browse More Insights of Towards Healthcare:
The global compound management market size is calculated at US$ 492.14 million in 2024, grew to US$ 563.56 million in 2025, and is projected to reach around US$ 1899.41 million by 2034. The market is expanding at a CAGR of 14.51% between 2025 and 2034.
The compounded topical drug market was estimated at US$ 4.27 billion in 2023 and is projected to grow to US$ 6.78 billion by 2034, rising at a compound annual growth rate (CAGR) of 4.29% from 2024 to 2034.
The global compounding pharmacy market size was estimated at US$ 13.19 billion in 2023 and is projected to grow to US$ 22.91 billion by 2034, rising at a compound annual growth rate (CAGR) of 5.15% from 2024 to 2034.
The global compounding chemotherapy market size is calculated at US$ 5.34 billion in 2024, grew to US$ 5.68 billion in 2025, and is projected to reach around US$ 9.97 billion by 2034. The market is expanding at a CAGR of 6.44% between 2025 and 2034.
The global vials used in compounding pharmacy market is surging, with an overall revenue growth expectation of hundreds of millions of dollars during the forecast period from 2024 to 2034. Projected revenue growth is set to skyrocket while the market held a notable share in 2025.
The global bulk-drug compounding market size was estimated at USD 12.52 billion in 2025 and is predicted to increase from USD 13.18 billion in 2026 to approximately USD 20.87 billion by 2035, expanding at a CAGR of 5.24% from 2026 to 2035.
The global animal drug compounding market size was estimated at USD 1.68 billion in 2025 and is predicted to increase from USD 1.82 billion in 2026 to approximately USD 3.73 billion by 2035, expanding at a CAGR of 8.32% from 2026 to 2035.
The global GCC compounding pharmacy market size is calculated at USD 17.91 million in 2025, grew to USD 19.57 million in 2026, and is projected to reach around USD 43.46 million by 2035. The market is expanding at a CAGR of 9.27% between 2026 and 2035.
The U.S. compounding pharmacy market size in 2025 was US$ 6.98 billion, expected to grow to US$ 7.42 billion in 2026 and further to US$ 12.79 billion by 2035, backed by a robust CAGR of 6.24% between 2026 and 2035.
The medical-grade polyvinyl chloride (PVC) compounds market size was estimated at USD 1.30 billion in 2025 and is predicted to increase from USD 1.37 billion in 2026 to approximately USD 2.15 billion by 2035, expanding at a CAGR of 5.13% from 2026 to 2035.
Segments Covered in the Report
By Product Type
- Sterile Compounded Drugs
- Parenteral formulations
- IV solutions
- IV injections
- Infusions (nutritional & therapeutic)
- Ophthalmic sterile products
- Injectable antibiotics
- Sterile hormonal formulations
- Parenteral formulations
- Non-sterile Compounded Drugs
- Oral liquids and suspensions
- Topical creams and ointments
- Gels and lotions
- Capsules & tablets (customized strength/dosage)
- Lozenges and troches
By Formulation Type
- Injections & Infusions
- Single-dose vials
- Multi-dose vials
- Pre-filled syringes
- Nutritional IV therapy (parenteral nutrition)
- Solutions
- Oral solutions
- Topical solutions
- Ophthalmic solutions
- Suspensions
- Oral suspensions
- Injectable suspensions
- Pediatric suspensions
- Creams & Gels
- Hormonal creams/gels (HRT)
- Pain relief gels/creams
- Dermatological creams (eczema, psoriasis, acne)
- Capsules & Tablets
- Customized strength oral capsules
- Compounded tablets for chronic conditions
- Pediatric dosage forms
By Therapeutic Application
- Pain Management
- Chronic pain management
- Post-surgical pain
- Neuropathic pain
- Hormone Replacement Therapy (HRT)
- Bioidentical hormone therapy
- Estrogen, progesterone, testosterone formulations
- Menopausal symptom relief
- Oncology
- Chemotherapy supportive care drugs
- Anti-nausea and pain relief for cancer patients
- Ophthalmology
- Eye drops for glaucoma, dry eyes
- Post-surgical ophthalmic solutions
- Infectious Disease Treatment
- Injectable antibiotics
- Antifungal preparations
- Antiviral formulations
- Cardiology
- Injectable cardiac drugs
- Oral compounded medications for heart conditions
- Gastroenterology
- Digestive enzyme formulations
- Oral suspensions for GI disorders
- Nutritional Infusions
- Parenteral nutrition (PN)
- Vitamins and micronutrient infusions
- Pediatrics
- Pediatric oral solutions
- Custom dosing formulations
- Dermatology
- Topical creams, gels, and ointments
- Anti-aging and cosmetic compounds
- Treatment for skin disorders
By Compounding Type
- Batch Compounding
- Large-scale sterile compounding for hospitals/clinics
- Standardized formulations for recurring prescriptions
- Patient-specific Compounding
- Customized dose per patient requirement
- Pediatric formulations with specific dosing
- Oncology and HRT patient-specific prescriptions
By Country
- Northeast
- Midwest
- South
- West
Immediate Delivery Available | Buy This Research Report Now @ https://www.towardshealthcare.com/checkout/5386
About Us
Towards Healthcare is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region: +44 778 256 0738
North America Region: +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Our Trusted Data Partners
Precedence Research | Towards Packaging | Towards Food and Beverages | Towards Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest



